|
The students explored some of the properties of eggs this week, including how lecithin, a fat like molecule (phospholipid) found in the yolk can serve as an emulsifier, bringing together oil and water mixtures because of its hydrophilic head and hydrophobic tail.
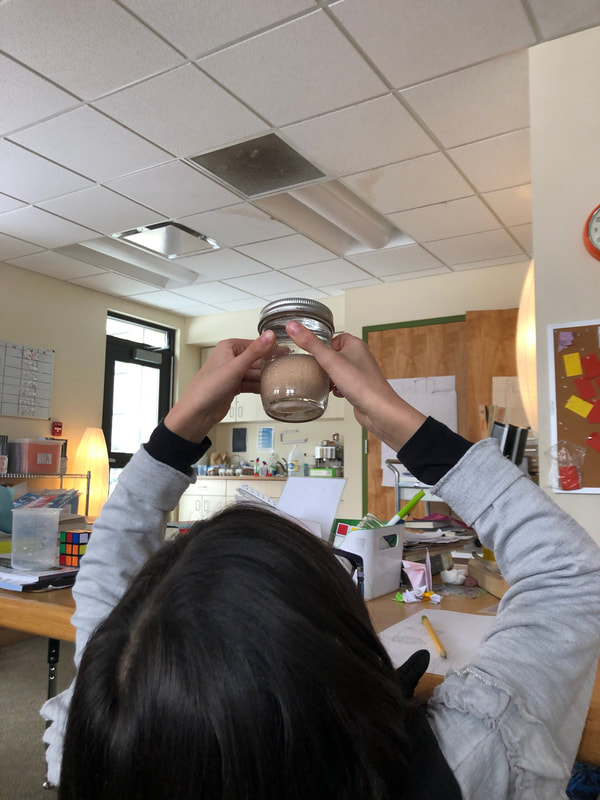
We began by observing how water and oil interact (or don't) in mason jars in small groups. After shaking them the students noticed that the oil and water immediately separated. We then watched this video which explains why water and oil don't mix. The video used the metaphor of dancers on a dance floor. Another video explained how lecithin works as an emulsifier. The structure of the lecithin forms a barrier that prevents the fat droplets from coming into contact with other droplets. With this knowledge in hand, the students were asked to tell a short story through images and/or words, creating characters for oil, water and lecithin and describing how lecithin was ultimately able to bring the two together. We also added egg yolks to our initial oil/water mixtures but noticed no matter how hard we shook them, the water and oil would separate. Thinking we needed more yolks, we continued to add but the results were the same. The next day, we combined the mixtures with a small hand blender and our emulsion was complete.
0 Comments
This week the students conducted The Naked Egg experiment. In small groups, they immersed an egg in vinegar, letting it sit for 7 days, changing the vinegar in the jars once. When the egg was first exposed to the vinegar, they noticed bubbles forming on the surface of the eggshell. After, they were asked to make predictions about what they thought would happen. Once the week passed, we removed the eggs from the vinegar and observed the changes. They noticed that the shell had dissolved and that the egg itself felt rubbery, like a bouncy ball. It was also translucent.
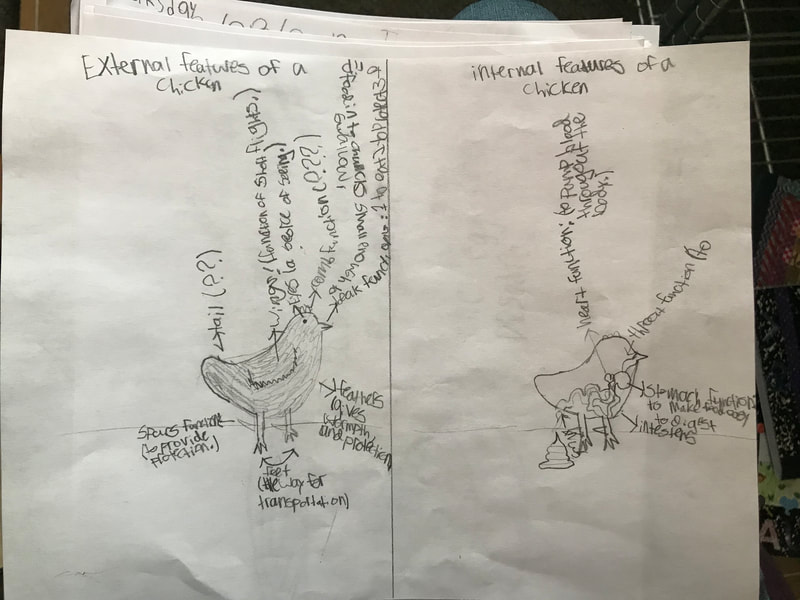
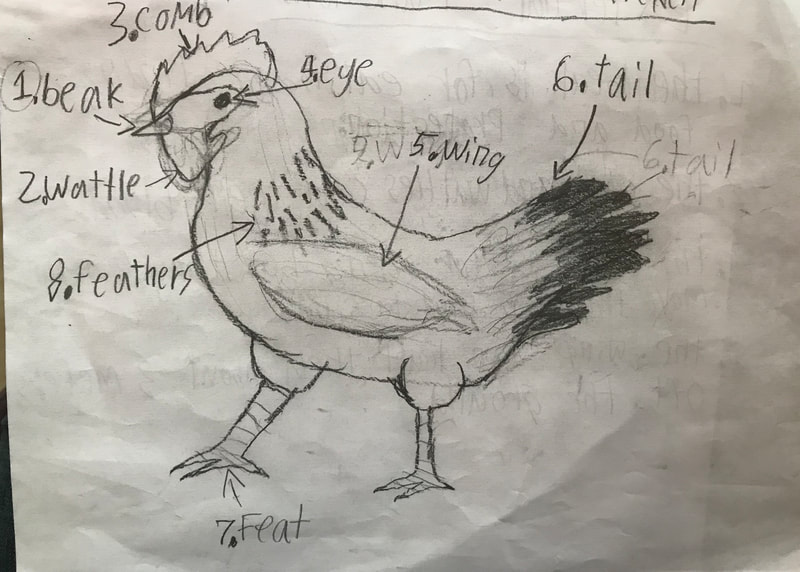
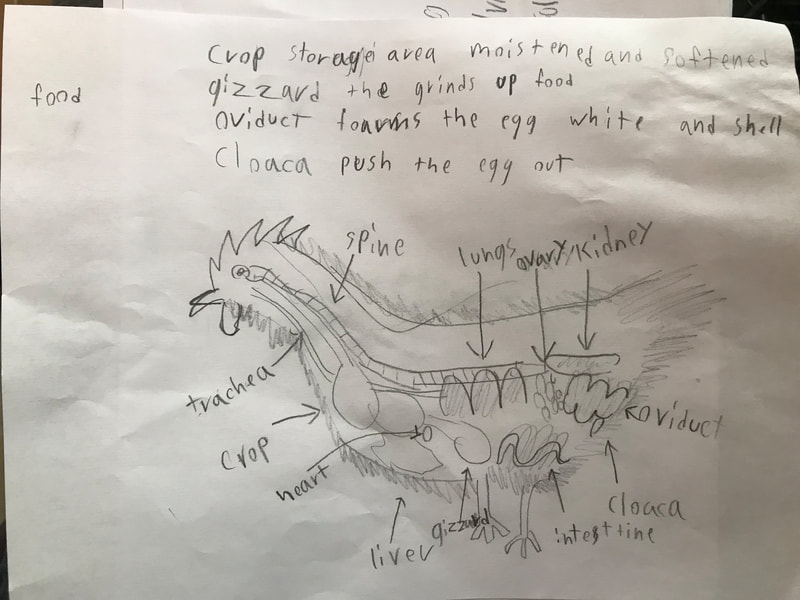
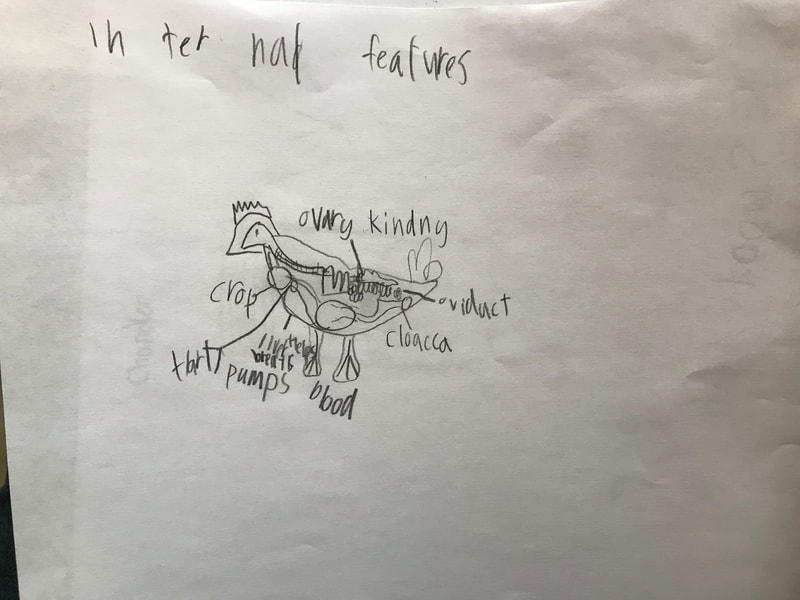
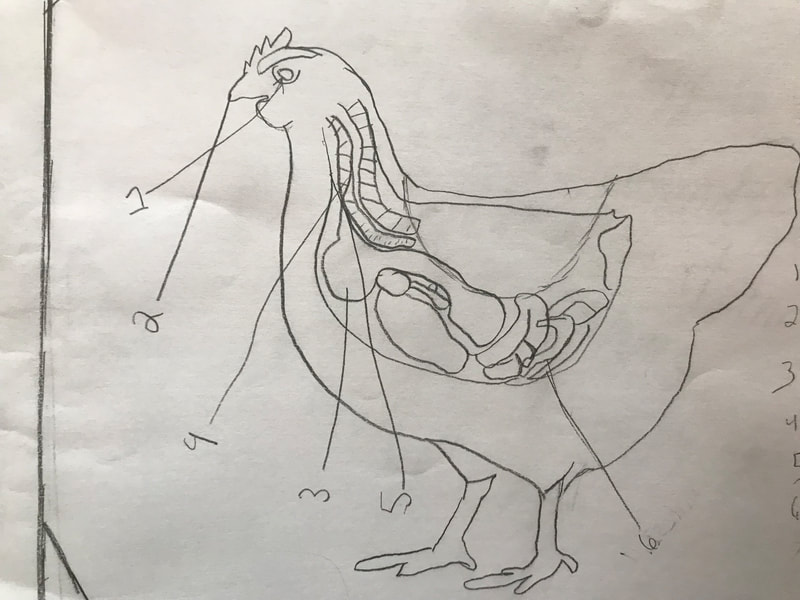
We discussed the difference between a physical reaction and a chemical reaction as well as the specifics about what occurred here, with the acetic acid from the vinegar reacting with the calcium carbonate of the shell, dissolving it. The reaction also produced water and carbon dioxide, the latter being the bubbles they saw on the surface of the egg. The excitement surrounding the changes prompted them to propose further experiments involving eggs and a variety of other substances including varying the strength of the acids or parts of the egg used in the experiment. This semester we will be digging into the science behind all things chicken! We began the project by researching what we call "the recipe of a chicken". We don't mean this in terms of how to prepare a chicken for dinner. Rather, if students had to compose a chicken from scratch (think: Franken-chicken), what are all the parts, inside and out, that they need to include?
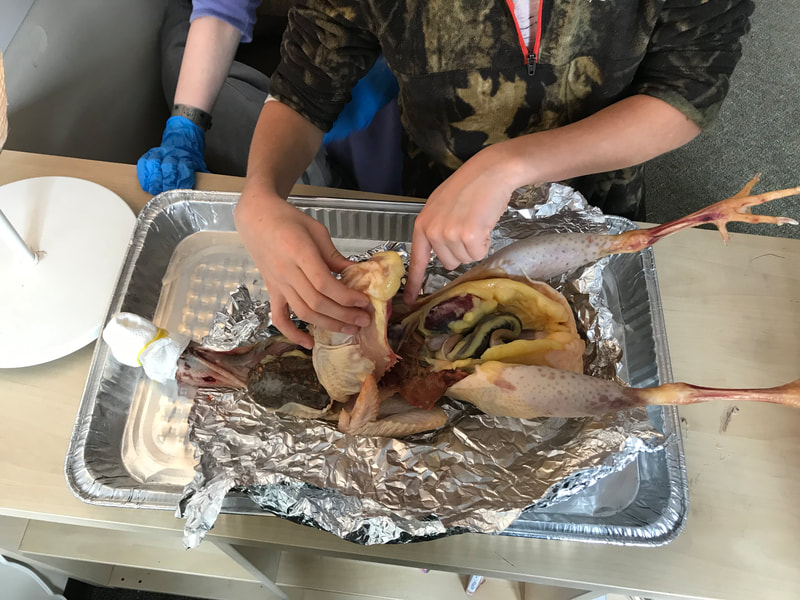
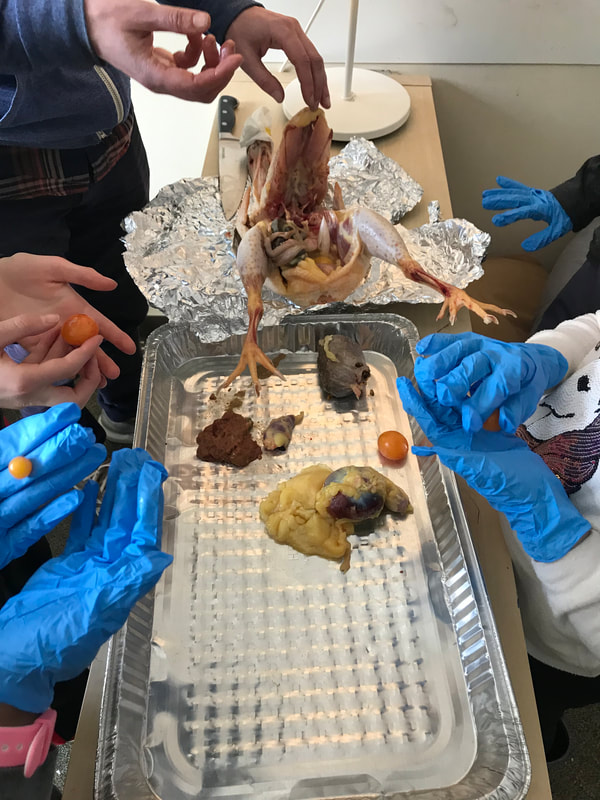
First, students spent time researching the internal/external features of the bird and illustrating these. Next, students were able to connect their research to the real thing through a hands-on exploration! Thanks to Mary Perrin and Jean Marie Rouillard for sharing their birds with us! Students absolutely loved this activity. We were able to trace the digestion tracks, hold chicken lungs, and even see eggs in various stages of development with the shells still in the process of forming. Finally, Jill Keller, SK parent and Clinical Assistant Professor in the Unit for Laboratory Animal Medicine at the University of Michigan, joined the class to discuss more in-depth, specific anatomical systems of the chicken, such as the skeletal system and digestion system. |
Archives
May 2019
Categories |
















































 RSS Feed
RSS Feed
